Abstract
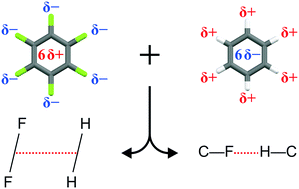
The evolution of the structure of the four solid phases of the prototype binary-adduct C6H6 : C6F6 as a function of temperature has been investigated using X-ray and neutron diffraction. An explanation is proposed concerning changes in the arrangements of the molecules at each of the three phase transitions and the dynamics in C6H6 : C6F6 are briefly compared with those of the adduct formed between mesitylene and C6F6. The observations are rationalised using simple models of intermolecular electrostatics.


 Please wait while we load your content...
Please wait while we load your content...