Three novel Co(ii)/Ni(ii)-based coordination polymers as efficient heterogeneous catalysts for dye degradation†
Abstract
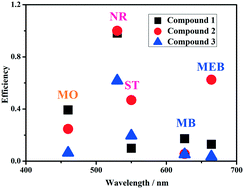
Three novel networks, namely, {[Co(HL)2]}n (1), {[Co(L)(H2O)]·[Co3/2(L)(OH-Hbdc)(H2O)2]·H2O}n (2), and [Ni(L)(H2O)5]·DMF·4H2O (3), where H2L = 4′-pyridin-4-yl-biphenyl-3,5-dicarboxylic acid and OH-H2bdc = 5-hydroxyisophthalic acid, have been hydrothermally synthesized and characterized. Compound 1 is a two-dimensional (2D) layered structure of Co2+ ions coordinated with incompletely deprotonated HL− ligands. Compound 2 is a crystal in the triclinic space group P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) and shows a very rare 2D + 2D heterogeneous framework built from 3-connected {63} hcb layers and 4-connected {44·62} sql layers. Compound 3 is a 0-D molecular complex crystallized in the chiral space group P212121, which suggests that symmetry breaking can be driven by the solvent-assisted homochiral helix without any chiral building blocks. In addition, their heterogeneous catalytic oxidation activities toward methyl orange (MO), methyl blue (MB), neutral red (NR), methylene blue (MEB), and safranine T (ST) and the mechanisms in the presence of H2O2 have been investigated in detail in aqueous solutions. The results show that the photocatalytic efficiency of NR is the highest for compounds 1–3.
and shows a very rare 2D + 2D heterogeneous framework built from 3-connected {63} hcb layers and 4-connected {44·62} sql layers. Compound 3 is a 0-D molecular complex crystallized in the chiral space group P212121, which suggests that symmetry breaking can be driven by the solvent-assisted homochiral helix without any chiral building blocks. In addition, their heterogeneous catalytic oxidation activities toward methyl orange (MO), methyl blue (MB), neutral red (NR), methylene blue (MEB), and safranine T (ST) and the mechanisms in the presence of H2O2 have been investigated in detail in aqueous solutions. The results show that the photocatalytic efficiency of NR is the highest for compounds 1–3.


 Please wait while we load your content...
Please wait while we load your content...