Acidochromic spiropyran–merocyanine stabilisation in the solid state†
Abstract
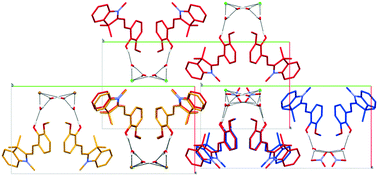
In this work, we present an innovative approach to stabilise the open-ring merocyanine form of a spiropyran compound. In the solid state, the merocyanine form is rarely observed, highlighting the importance of this contribution. Our concept is based on a co-crystallisation approach of the open form with an inorganic acid. This way, we identified a reliable technique for stabilising the coloured merocyanine form, and in one specific case, we even obtained crystals with increased stability towards photodegradation. 15 novel crystal structures of spiropyran derivatives with several inorganic acids were determined by single crystal X-ray diffraction. Solvent evaporation of spiropyran compounds, i.e. 1,3,3-trimethylindolinobenzopyrylospiran (SPH), 1,3,3-trimethylindolino-β-naphthopyrylospiran (SPBenz), 1,3,3-trimethylindolino-8′-methoxybenzopyrylospiran (SPOMe) and 1,3,3-trimethylindolino-6′-nitrobenzopyrylospiran (SPNO2), in the presence of inorganic acids, namely hydrochloric acid, hydrobromic acid, nitric acid, sulphuric acid and phosphoric acid, yielded the rare open-ring merocyanine form stabilised by protonation of the primary oxygen atom unfolded by ring-opening isomerisation and compensating the charge with a hydrogen bonded anion. Analysis of the solid state properties was performed by thermal gravimetric analysis and diffuse reflectance spectroscopy.


 Please wait while we load your content...
Please wait while we load your content...