Pillars of crystal engineering: crystal energies and symmetry operators†
Abstract
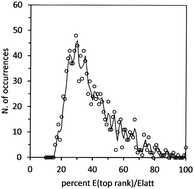
Interaction energies in molecular systems endowed with three-dimensional long range symmetry, i.e. crystals, are electromagnetic in nature and can be calculated by quantum mechanical methods. Approximate schemes are also available. Total lattice energies are compared, with some restriction, to sublimation enthalpies and are useful for calibration purposes, but they carry little structural, analytical or predictive information. Pairwise molecular interactions in crystals reveal the relative importance of single recognition modes and at the same time, rank the importance of symmetry operators. By analyzing the crystal packing and lattice energies of 1235 organic crystals, along with their breakdown into molecule–molecule interaction energies, we show that large differences in the kinds and roles of top-ranking molecule–molecule interactions appear in crystals as a function of molecular size, chemical constitution, chemical functionalities, hydrogen-bonding abilities, and number of molecules in the asymmetric unit. Top-ranking molecular pairs never exceed 20% of the total lattice energy. Like all models, this bimolecular reductionist approach has its limitations that are highlighted along with its merits.
- This article is part of the themed collection: CrystEngComm 20th volume collection


 Please wait while we load your content...
Please wait while we load your content...