Probing semiconductivity in crystals of stable semiquinone radicals: organic salts of 5,6-dichloro-2,3-dicyanosemiquinone (DDQ) radical anions†
Abstract
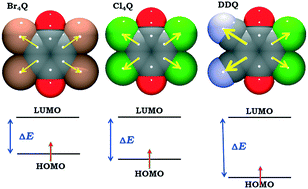
The structural parameters and semiconductivity of crystals with stacked 5,6-dichloro-2,3-dicyanosemiquinone (DDQ) radicals were studied for a series of the first nine salts of DDQ with substituted N-ethyl- and N-methylpyridinium cations. Structures with stacks of equidistant radicals (interplanar separation <3.3 Å) are fairly good semiconductors (10−9 S cm−1), while those with Peierls-distorted stacks comprising diamagnetic dimers of radicals (interplanar separations are <3.1 Å within the dimers and >3.4 Å between the dimers) are insulators. DFT calculations indicate the covalent character (pancake bonding) of the interactions between the DDQ radical anions. The inductive effects of electronegative substituents (halogeno and cyano) on the quinoid ring stabilise the radicals, but also increase the HOMO–LUMO gap, resulting in lower electrical conductivity.


 Please wait while we load your content...
Please wait while we load your content...