Electrochemical performance and structure of Al2W3−xMoxO12†
Abstract
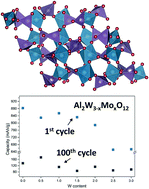
The A2M3O12 class of materials are mostly known for their negative thermal expansion properties, however, little research has been conducted in regards to their electrochemical properties. The A2M3O12 structure consists of rigid polyhedra, featuring large structural voids which may be sufficiently large to reversibly host Li-, Na- and K-ions with minimal strain being induced during insertion/extraction. In the present work, members of the Al2W3−xMoxO12 (0 ≤ x ≤ 3) family were synthesised, and characterised structurally and electrochemically. The Al2W3−xMoxO12 solid solutions adopt a monoclinic P21/a space-group symmetry which transforms to the orthorhombic Pnca space-group symmetry between the compositions Al2W2MoO12 and Al2W2.5Mo0.5O12. The W and Mo cations appear to be randomly distributed on the 6 or 2 independent crystallographic sites in the monoclinic or orthorhombic symmetries, respectively. Discharge against Li yields a relatively high first discharge capacity of 883 mA h g−1 for Al2Mo3O12 with a reversible capacity of 136 mA h g−1 by the 100th cycle which seems to be relatively insensitve to the applied currents used here. The capacities decrease with increasing W content to 654 mA h g−1 for Al2W3O12 in the first and 96 mA h g−1 in the 100th discharge cycle. X-ray absorption spectroscopy and ex situ synchrotron X-ray diffraction data were used to describe the change in oxidation state of Mo and the structural evolution upon discharge and indicate the formation of an amorphous or nanocrystalline phase(s) upon lithiation. Na- and K-half-cells achieved reversible discharge capacities below 28 mA h g−1 while typically introducing less change in the structure. This work illustrates the influence of composition and structure on the electrochemical performance in this family of compounds.


 Please wait while we load your content...
Please wait while we load your content...