On-surface nickel porphyrin mimics the reactive center of an enzyme cofactor†
Abstract
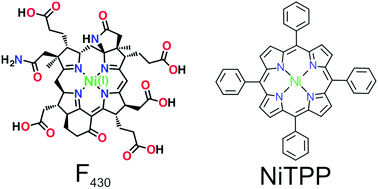
Metal-containing enzyme cofactors achieve their unusual reactivity by stabilizing uncommon metal oxidation states with structurally complex ligands. In particular, the specific cofactor promoting both methanogenesis and anaerobic methane oxidation is a porphyrinoid chelated to a nickel(I) atom via a multi-step biosynthetic path, where nickel reduction is achieved through extensive molecular hydrogenation. Here, we demonstrate an alternative route to porphyrin reduction by charge transfer from a selected copper substrate to commercially available 5,10,15,20-tetraphenyl-porphyrin nickel(II). X-ray absorption measurements at the Ni L3-edge unequivocally show that NiTPP species adsorbed on Cu(100) are stabilized in the highly reactive Ni(I) oxidation state by electron transfer to the molecular orbitals. Our approach highlights how some fundamental properties of synthetically inaccessible biological cofactors may be reproduced by hybridization of simple metalloporphyrins with metal surfaces, with implications towards novel approaches to heterogenous catalysis.


 Please wait while we load your content...
Please wait while we load your content...