Palladium triggered diene formation from nitro allylic compounds: a versatile entry into naphthalene derivatives†
Abstract
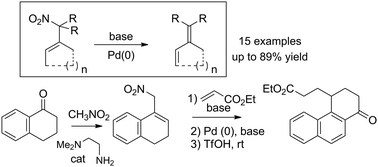
Nitro allylic derivatives were converted into dienes through the elimination of the nitro group under basic treatment, in the presence of a palladium catalyst. This reaction probably involves the formation of a palladium π-allyl complex followed by a base-promoted β-hydride elimination. This reaction, combined with the condensation of ketones with nitromethane and the functionalization of the resulting nitrocycloalkenes, constitutes a very powerful synthetic tool for the formation of dienes. Particular attention has been brought to the application of this methodology to the formation of 1-substituted naphthalenes from 1-tetralone.


 Please wait while we load your content...
Please wait while we load your content...