Efficient and durable N2 reduction electrocatalysis under ambient conditions: β-FeOOH nanorods as a non-noble-metal catalyst†
Abstract
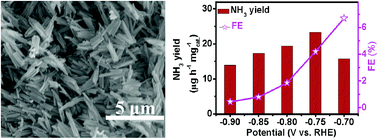
NH3 is one of the most important chemicals with a wide range of applications. NH3 is mainly produced via the Haber–Bosch process, which leads to large energy consumption and carbon emission. Electrochemical reduction has emerged as an environmentally-benign and sustainable alternative for artificial N2 fixation under ambient conditions, but needs materials to effectively catalyze the N2 reduction reaction (NRR). In this communication, we report that β-FeOOH nanorods behave as an efficient and durable NRR electrocatalyst. In 0.5 M LiClO4, such an electrocatalyst achieves a high NH3 yield of 23.32 μg h−1 mgcat.−1 and a faradaic efficiency of 6.7%, outperforming most reported aqueous-based NRR electrocatalysts under ambient conditions. Notably, this catalyst also shows good electrochemical stability and excellent selectivity. The catalytic mechanism of the NRR on the FeOOH(110) surface is further discussed using density functional theory calculations.


 Please wait while we load your content...
Please wait while we load your content...