A naphthoimidazolium-cholesterol derivative as a ratiometric fluorescence based chemosensor for the chiral recognition of carboxylates†
Abstract
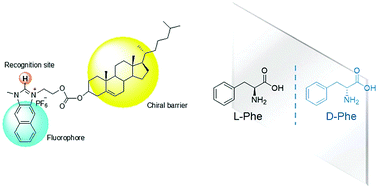
Fluorescent chemosensors for sensing chiral molecules have been actively studied in recent years. In the current study, we report a naphthoimidazolium-cholesterol derivative (NI-chol 1) as a fluorescence based chemosensor for chiral recognition, in which the naphthoimidazolium serves not only as a fluorophore but also as a recognition moiety for anions via imidazolium (C–H)+–anion binding and the cholesterol unit acts as a chiral barrier. In particular, NI-chol 1 displayed unique and distinct ratiometric changes with Boc-D-Phe, on the other hand, Boc-L-Phe induced a negligible change. Furthermore, a distinct downfield shift (from 9.64 ppm to 9.96 ppm) of the imidazolium C–H peak was observed for Boc-D-Phe (5 eq.) with severe broadening, which indicates strong ionic hydrogen bonding between the C–H proton and the carboxylate.
- This article is part of the themed collection: The Mechanics of Supramolecular Chemistry


 Please wait while we load your content...
Please wait while we load your content...