Dual role of ethyl bromodifluoroacetate in the formation of fluorine-containing heteroaromatic compounds†
Abstract
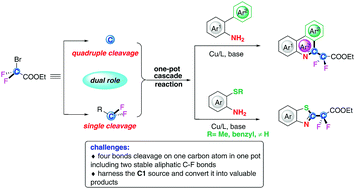
An efficient one-pot cascade process via unprecedented quadruple cleavage of BrCF2COOEt with primary amines to afford valuable fluorine-containing heterocycles is described, in which BrCF2COOEt plays a dual role as a C1 synthon and a difluoroalkylating reagent for the first time. Mechanistic studies supported by DFT calculations suggest that a base plays an active role in the formation of the key intermediate isocyanides generated in situ from primary amines and difluorocarbene.


 Please wait while we load your content...
Please wait while we load your content...