An anion receptor that facilitates transmembrane proton–anion symport by deprotonating its sulfonamide N–H proton†
Abstract
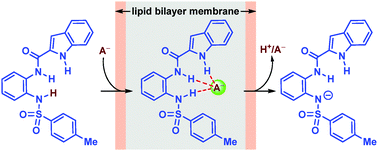
Indole-based amide-sulfonamide derivatives were synthesized. The X-ray crystal structure and chloride binding studies in solution showed a 1 : 1 stoichiometry. The ion transport efficiency directly correlated to the pKa of the sulfonamide N–H protons. The mechanistic study indicated proton–anion symport across the lipid bilayer membrane.


 Please wait while we load your content...
Please wait while we load your content...