Highly enantioselective synthesis of trifluoromethyl cyclopropanes by using Ru(ii)–Pheox catalysts†
Abstract
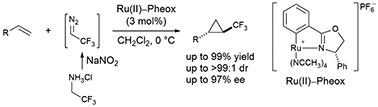
An asymmetric synthesis of various trifluoromethyl cyclopropanes from olefins, such as vinyl ferrocene, vinyl ethers, vinyl amines, vinyl carbamates and dienes, was achieved by using Ru(II)–Pheox catalysts. This catalytic system can function at a low catalyst loading (3 mol%) compared with those reported previously, and the desired cyclopropane products are obtained in high yields with excellent diastereoselectivity (up to >99 : 1) and enantioselectivity (up to 97% ee).


 Please wait while we load your content...
Please wait while we load your content...