P-glycoprotein targeted photodynamic therapy of chemoresistant tumors using recombinant Fab fragment conjugates†
Abstract
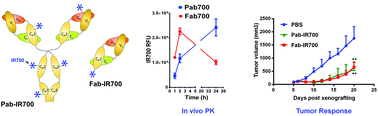
P-glycoprotein (Pgp) has been considered as a major cause of cancer multidrug resistance; however, clinical solutions to overcome this drug resistance do not exist despite the tremendous endeavors. The lack of cancer specificity is a main reason for clinical failure of conventional approaches. Targeted photodynamic therapy (PDT) is highly cancer specific by combining antibody targeting and locoregional light irradiation. We aimed to develop Pgp-targeted PDT using antibody–photosensitizer conjugates made of a recombinant Fab fragment. We prepared the photosensitizer conjugates by expressing a recombinant Fab fragment and specifically linking IR700-maleimide at the C-terminal of the Fab heavy chain. In vitro studies showed that the Fab conjugates specifically bind to Pgp. Their phototoxicity was comparable to full antibody conjugates when assayed with conventional 2-D cell culture, but they outperformed the full antibody conjugates in a 3-D tumor spheroid model. In a mouse xenograft model of chemoresistant tumors, Fab conjugates showed Pgp specific delivery to chemoresistant tumors. Upon irradiation with near-infrared light, they caused rapid tumor shrinkage and significantly prolonged the survival of tumor-bearing mice. Compared to the full antibody conjugates, Fab conjugates took a shorter time to reach peak tumor levels and achieved a more homogeneous tumor distribution. This allows light irradiation to be initiated at a shorter time interval after the conjugate injection, and thus may facilitate clinical translation. We conclude that our targeted PDT approach provides a highly cancer-specific approach to combat chemoresistant tumors, and that the conjugates made of recombinant antibody fragments are superior to full antibody conjugates for targeted PDT.


 Please wait while we load your content...
Please wait while we load your content...