Photo-triggered micelles: simultaneous activation and release of microtubule inhibitors for on-demand chemotherapy†
Abstract
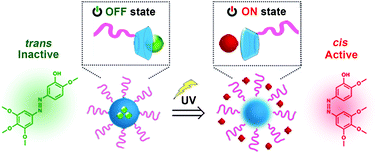
The nonspecific biodistribution of cytotoxic drugs and associated adverse effects greatly limit the efficacy and patient compliance of chemotherapy. To address this, we employed a photoswitchable microtubule inhibitor (Azo-CA4) that was physically loaded in cyclodextrin-bearing micellar nanocarriers through the host–guest interaction. Azo-CA4 was only activated upon ultraviolet (UV) light irradiation to trigger the transition from the “trans” (inactive) to “cis” (active) state. Such conformation change could then induce rapid Azo-CA4 release from micelles without the delay of the onset of therapeutic action. This nanoscale delivery system produced photo-triggered antimitotic and pro-apoptotic effects in MDA-MB-231 cells via a triggered control of microtubule dynamics. The anticancer efficacy of Azo-CA4-loaded micelles was further proved in vivo using a 4T1 tumor-bearing mice model coupled with multiple topical administrations to avoid the penetration problem of UV light. This work provides a new delivery vehicle to aid the application and potential translation of Azo-CA4 as biomedical tools and precision chemotherapeutics.


 Please wait while we load your content...
Please wait while we load your content...