Rapid screening of antimalarial drugs using low-temperature plasma desorption/ionization Orbitrap mass spectrometry†
Abstract
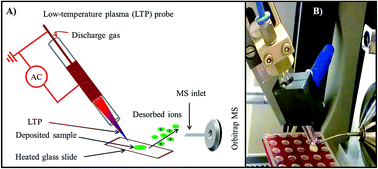
Antimalarial drugs for the treatment of malaria are amongst the most counterfeit drugs in third world countries. An analytical technique capable of rapid determination of active pharmaceutical agents in real drug samples with minimal sample preparation is a useful tool for quality control in the pharmaceutical industry, in the search for illicit drugs, and consumer safety. Low-temperature plasma mass spectrometry (LTP-MS) has the ability to carry out rapid analysis of samples in their native form with minimal sample preparation and is a promising tool for qualitative screening. In this study, a direct analysis method for the characterization of active ingredients in Coartem (artemether, lumefantrine) and Malarone (atovaquone, proguanil hydrochloride) tablets using a high-resolution mass spectrometer (HR-MS) and a home-built LTP desorption/ionization source was developed and optimized. Direct tablet analysis is completed in less than three minutes and does not require extensive sample preparation or the use of solvents. This is considered a significant benefit compared to spray-based ambient desorption/ionization methods or even a conventional high-performance liquid chromatography (HPLC) approach. Artemether and lumefantrine in Coartem were detected directly both as their protonated molecules in LTP-HR-MS. Atovaquone was detected after solvent extraction of Malarone. In addition, LTP-HR-MS provided characteristic fragment ions of the target molecules that aided in their structural identification even in the presence of a tablet matrix. LTP-HR-MS results were validated by HPLC-MS analysis of the samples under investigation.


 Please wait while we load your content...
Please wait while we load your content...