A direct electron transfer biosensor based on a horseradish peroxidase and gold nanotriangle modified electrode and electrocatalysis
Abstract
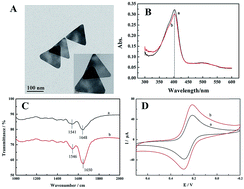
Gold nanotriangles (AuNTs) were used as a modifier on the surface of a carbon ionic liquid electrode (CILE), which was applied to realize direct electrochemistry and electrocatalysis of horseradish peroxidase (HRP). The modified electrode exhibited excellent activity for electrocatalytic reduction of trichloroacetic acid (TCA) and sodium nitrite (NaNO2). The presence of AuNTs on the electrode surface can bridge the gaps between the electrochemically active sites of HRP and substrate interfaces with the increase of the loading amount of HRP. TEM results confirmed the typical triangular shape of AuNTs and spectroscopic results revealed that no denaturation occurred in the structure of HRP molecules after they were mixed with AuNTs. The electrochemical behavior of Nafion/HRP/AuNTs/CILE was examined in pH 3.0 phosphate buffer solution with the kinetic effect being investigated. The apparent heterogeneous electron transfer rate constant (ks) and the charge transfer coefficient (α) were found to be 1.01 s−1 and 0.49. Electrocatalysis of TCA and NaNO2 was examined with the detection limits calculated to be 0.33 mmol L−1 and 0.53 mmol L−1, respectively. The proposed method was applied to a medical facial peel solution for TCA detection with satisfactory results.
- This article is part of the themed collection: Analytical Methods Recent HOT articles


 Please wait while we load your content...
Please wait while we load your content...