Novel, sensitive and selective colorimetric detection of arsenate in aqueous solution by a Fenton-like reaction of Fe3O4 nanoparticles
Abstract
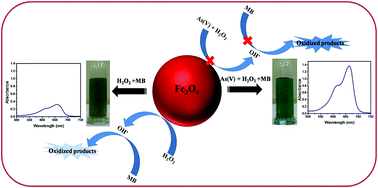
In this paper, we present a novel colorimetric sensor for the detection of arsenate in aqueous solution. Magnetic nanoparticles (Fe3O4 NPs) prepared by a hydrothermal process were characterized by SEM, XRD, FT-IR, and VSM analysis. The Fenton-like catalytic reaction of Fe3O4 NPs promoted the oxidation of methylene blue indicator (MB) in the presence of H2O2 which resulted in the diminishing of the dark blue colour of the solution, due to generation of ·OH radicals in the solution. This can be viewed by the naked eye and monitored by UV-Vis spectroscopy at 664 nm. Upon addition of arsenate to this system the blue colour regenerated in solution due to the agglomeration of As(V) on Fe3O4 which may block the active sites of the Fe3O4 NPs and decrease their intrinsic catalytic activity. Under optimal conditions, the proposed colorimetric sensor exhibited rapid and selective detection of As(V) in aqueous solution with a detection limit of 0.358 nM with a linear relationship with R2 = 0.997. To the best of our knowledge, this is the first report where arsenates can be detected colorimetrically by this sensing system (Fe3O4/MB/H2O2). The designed colorimetric strategy may provide a promising alternative method for detection of arsenates in real water samples.
- This article is part of the themed collection: Analytical Methods Recent HOT articles


 Please wait while we load your content...
Please wait while we load your content...