Advancement of analytical modes in a multichannel, microfluidic droplet-based sample chopper employing phase-locked detection†
Abstract
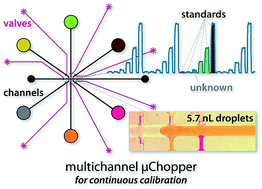
In this work, we expand upon our recently developed droplet-based sample chopping concepts by introducing a multiplexed fluidic micro-chopper device (μChopper). Six aqueous input channels were integrated with a single oil input, and each of these seven channels was controlled by a pneumatic valve for automated sampling through software control. This improved design, while maintaining high precision in valve-based droplet generation at bandwidths of 0.03 to 0.05 Hz, enabled a variety of analytical modes to be employed on-chip compared to previous devices limited to sample/reference alternations. The device was analytically validated for real-time, continuous calibration with a single sample and five standards; multiplexed analysis during calibration using a mixed mode; and standard addition through spiking of six sample droplets with varying amounts of standard. Finally, the standard addition mode was applied to protein quantification in human serum samples using on-chip, homogeneous fluorescence immunoassays. Ultimately, with only ∼1.2 μL of total analyzed solution volume—representing 100-fold and 75-fold reductions in reagent and serum volumes, respectively—we were able to generate full, six-point standard addition curves in only 1.5 min, and results correlated well with those from standard plate-reader equipment. This work thus exploited microfluidic valves for both their automation and droplet phase-locking capabilities, resulting in a micro-analytical tool capable of complex analytical interrogation modes on sub-microliter sample volumes while also leveraging drastic noise rejection via lock-in detection. The multichannel μChopper device should prove particularly useful in analyzing precious biological samples or for dynamic analyses at small volume scales.
- This article is part of the themed collections: Analytical Methods Recent HOT articles and Microfluidic systems with societal impact


 Please wait while we load your content...
Please wait while we load your content...