Controllable synthesis of cellulose benzoates for understanding of chiral recognition mechanism and fabrication of highly efficient chiral stationary phases†
Abstract
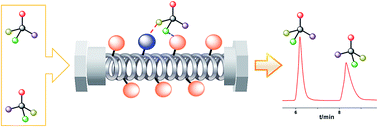
For understanding chiral recognition mechanism and fabricating high-efficient chiral stationary phases, the homogeneous benzoylation of cellulose with 8 benzoyl chloride derivatives was investigated in detail by using 1-ally-3-methylimizolium chloride as the solvent and pyridine as the catalyst. The reactivity of benzoyl chloride derivatives bearing different substituents was determined by the electronic nature of substituents, i.e., the linear Hammett relationship with a positive slope. Benzoyl chloride derivatives with stronger electron-withdrawing substituents exhibited higher reactivity. Taking advantage of the above reaction rules and excellent controllability of the homogeneous derivatization of cellulose, we synthesized 24 cellulose benzoates with different substituents and degree of substitution values. Then, the corresponding chiral stationary phases (CSPs) were fabricated using these cellulose benzoates as chiral selectors. The enantioseparation capability of CSPs was evaluated by high performance liquid chromatography with ten enantiomers. The enantioseparation results demonstrated that the hydrophobic helical conformation of cellulose derivatives was essential to the separation of racemates. Furthermore, due to some weak non-enantioselective interactions originating from a small amount of hydrogen-bonding donors (e.g., OH, NH), the corresponding CSPs exhibited a better and/or more universal enantioseparation capability. Based on the understanding of the chiral resolution mechanism, four novel, highly efficient and versatile CSPs were fabricated by controlling the chemical structure of the cellulose derivatives.
- This article is part of the themed collection: Analytical Methods Recent HOT articles


 Please wait while we load your content...
Please wait while we load your content...