A fast, direct, and sensitive analysis method for catechin determination in green tea by batch injection analysis with multiple-pulse amperometry (BIA-MPA)
Abstract
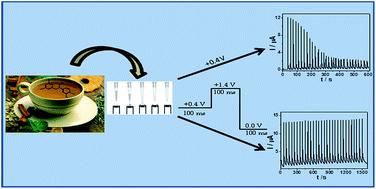
Bioactive compounds have several biological activities, promoting health. Among these compounds, those, such as vitamins and phenolic compounds, with antioxidant properties have attracted significant interest due to their proven effects in protection against oxidative stress. Most of the bioactive phenolic compounds are highly difficult to be determined by voltammetric techniques at bare electrodes since polymerization process occurs on the electrode surface during phenol oxidation. In this study, we described the determination of a bioactive compound, catechin, by electrochemical oxidation via batch injection analysis (BIA) with multiple amperometric detection (MPA) using an unmodified glassy carbon (GC) electrode in an acetate buffer solution (pH 4.5). The proposed BIA method is based on the application of multiple pulse potentials of 0.4 V (oxidation potential), 1.4 V (cleaning potential), and 0.0 V (conditioning potential) vs. Ag/AgCl (KCl 3 M), hence avoiding passivation by oxidation products and enabling the quantification of catechin. Under these conditions, the repeatability of the method for successive injections of catechin at a concentration of 50 μmol L−1 (n = 30) was 0.85%, and the sampling rate was 120 injections per hour. The amperometric response presented a linear range for solutions containing catechin at concentrations ranging from 20 to 100 μmol L−1 (R = 0.999), with a detection limit of 0.03 μmol L−1 and a quantification limit of 0.10 μmol L−1. In addition, the proposed method was shown to be fast and sensitive for the determination of catechin in real samples, and the results were comparable to those obtained via high-performance liquid chromatography (HPLC), at a 95% confidence level.


 Please wait while we load your content...
Please wait while we load your content...