A highly selective “turn-on” fluorescent sensor for zinc ion based on a cinnamyl pyrazoline derivative and its imaging in live cells†
Abstract
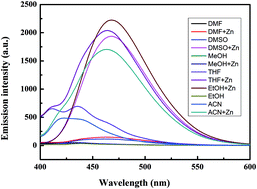
A fluorescence probe based on cinnamyl pyrazoline was developed and synthesized. The probe detected Zn2+ ion that was mixed with a series of cations in aqueous ethanol with high selectivity and high sensitivity. After adding Zn2+, the fluorescence intensity was significantly enhanced, with a 56-fold increase. The detection limits and binding constants of 2.95 × 10−8 M and 1.468 × 104 M−1 were calculated by a fluorescence titration experiment. The 1 : 1 binding chemometrics of the 1–Zn2+ complex was analyzed by Job's plot. The probe showed good reversibility when adding PPi. Additionally, the “turn-on” fluorescent probe with excellent membrane permeability was satisfactorily applied to microscopic imaging for the detection of Zn2+ in baby hamster kidney (BHK)-21 cells.


 Please wait while we load your content...
Please wait while we load your content...