Carbon nanodot aqueous binding phase-based diffusive gradients in thin films device for measurement of dissolved copper and lead species in the aquatic environment†
Abstract
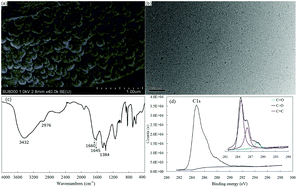
A reliable method for the determination of dissolved Cu2+ and Pb2+ species in water via a diffusive gradient in thin films (DGT) device using water-soluble carbon nanodots (CD) as the binding agent was developed (CD-DGT). The uptake of dissolved Cu2+ and Pb2+ species by CD-DGT increased linearly with a deployment time of over 120 h, while the uptake of Ca2+, Mg2+, Cd2+, Zn2+, Ni2+, and Co2+ by CD-DGT has no significant linear accumulation during the same deployment time. The concentrations of the dissolved Cu2+ and Pb2+ species measured using CD-DGT are in good agreement with the element concentrations spiked directly in a tested synthesis solution. The performance of CD-DGT for the determination of dissolved Cu2+ and Pb2+ species is found to be independent of ionic strength in the range of 0.001–0.05 mol L−1 and in the pH range from 5 to 8. The presence of fulvic acid and tannic acid has no significant influence on the uptake of dissolved Cu2+ and Pb2+ species by CD-DGT under the tested conditions. In different water samples, good agreement was observed between the dissolved Cu2+ and Pb2+ concentrations measured by CD-DGT, and diffusive equilibration in the thin film devices was found. Based on the results obtained, CD-DGT enables the selective and quantitative determination of dissolved Cu2+ and Pb2+ species in water.


 Please wait while we load your content...
Please wait while we load your content...