Tuning the electronic properties and the planarity degree in the π-extended TTF series: the prominent role of heteroatoms†
Abstract
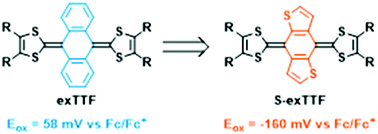
The main asset of small molecules for application in organic electronics lies in the tunability of their electronic properties owing to the precise control of their molecular design. Semiconducting properties in organic compounds are for instance closely linked to the molecular planarity degree, including when considering various redox states. Among those species, the π-extended TTF (exTTF: 9,10-bis(1,3-dithiol-2-ylidene)-9,10-dihydroanthracene) presents fascinating redox and structural properties, which have been extensively studied in various fields of organic electronics. Here we show that S-exTTF, a sulfur enriched π-isoelectronic dithieno analogue of pristine exTTF, synthesized through a Horner–Wadsworth–Emmons olefination, presents a much higher π-donating ability than its exTTF homologue, associated to a higher planarity degree in the neutral or in the oxidized states. This is demonstrated by a combined experimental/theoretical approach. Solution studies (cyclic voltammetry and thin layer cyclic voltammetry) as well as solid-state analyses (X-ray structures of neutral S-exTTF and of the electrocrystallized cation radical salt ([S-exTTF][PF6]·THF)) were performed, and systematically compared with those of the pristine exTTF analogue. An in-depth computational study, carried out on the neutral state as well as on oxidized states of S-exTTF, confirms the prominent role of intramolecular S⋯S interactions in dictating a planar conformation, which is manifested by exceptionally short S⋯S distances in the X-ray structure (2.82 Å, i.e., shorter, by far, than the sum of the van der Waals radii for S atoms (3.60 Å)), and in promoting HOMO destabilization compared to the parent exTTF.
- This article is part of the themed collection: Functional Organic Materials for Optoelectronic Applications


 Please wait while we load your content...
Please wait while we load your content...