Controlling the charge transfer flow at the graphene/pyrene–nitrilotriacetic acid interface†
Abstract
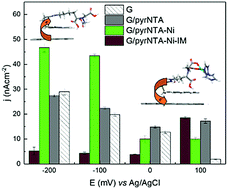
The fabrication of highly efficient bio-organic nanoelectronic devices is still a challenge due to the difficulty in interfacing the biomolecular component to the organic counterparts. One of the ways to overcome this bottleneck is to add a self-assembled monolayer (SAM) in between the electrode and the biological material. The addition of a pyrene–nitrilotriacetic acid layer to a graphene metal electrode enhances the charge transfer within the device. Our theoretical calculations and electrochemical results show that the formation of a pyrene–nitrilotriacetic acid SAM enforces a direct electron transfer from graphene to the SAM, while the addition of the Ni2+ cation and imidazole reverses the charge transfer direction, allowing an atomic control of the electron flow, which is essential for a true working device.


 Please wait while we load your content...
Please wait while we load your content...