Micellar chemotherapeutic platform based on a bifunctional salicaldehyde amphiphile delivers a “combo-effect” for heightened killing of MRSA†
Abstract
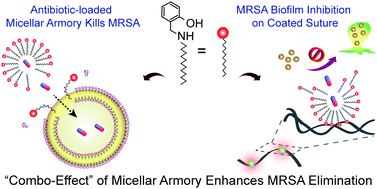
The devastating infections caused by methicillin-resistant Staphylococcus aureus (MRSA) coupled with its high resistance towards antibiotics underscores the need for an effective anti-MRSA therapeutic. The present study illustrates the use of a salicylaldehyde based bactericidal amphiphile (C1) in generating a micellar carrier that renders delivery of therapeutic antibiotics. The inherent membrane-targeting activity of C1 present in the micelle could be leveraged to counter the resistance of MRSA and enhance cellular uptake of the released antibiotics, resulting in effective elimination of the pathogen. The inherent bactericidal and antibiofilm activity of C1 was captured in FESEM analysis, solution-based assays and fluorescence microscopy. ANS-based fluorescence spectroscopy indicated that the critical micelle concentration (CMC) for C1 was 18.5 μM in water. DLS studies and FESEM analysis indicated that the average particle size for micelles based on C1 (C1M) and rifampicin-loaded C1M (C1M − R) was smaller than vancomycin-loaded C1M (C1M − V). C1M − R and C1M − V rendered sustained release of the antibiotics in physiologically relevant fluids. Notably, following interaction with MRSA for 3 h, the relative anti-MRSA activity of C1M − R and C1M − V was nearly 12-fold and 8-fold higher, respectively, as compared to the free antibiotics at equivalent concentration, highlighting the merit of leveraging the activity of C1 and the antibiotic concurrently in the micellar system. The relative cell-free antibiotic was also manifold lower in the case of C1M − R and C1M − V treated MRSA as against treatment with free antibiotics, suggesting that the amphiphilic warhead breached the membrane barrier and enhanced cellular uptake of the released antibiotics. Interestingly, C1M − R and C1M − V exhibited a high therapeutic index, being non-toxic to HEK 293 cells at concentrations higher than their minimum inhibitory concentration (MIC) against MRSA and they could be employed as an antibacterial coating to prevent MRSA biofilm formation on surgical silk sutures. The antibiotic-replete biocompatible micelles based on a self-assembling membrane-targeting amphiphile described herein represent a promising framework to integrate multiple warheads and generate a potent anti-MRSA therapeutic material.


 Please wait while we load your content...
Please wait while we load your content...