Interplay between the metal-support interaction and stability in Pt/Co3O4(111) model catalysts†
Abstract
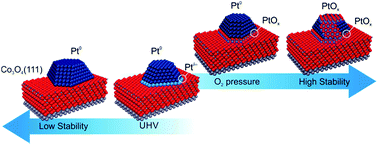
The interplay between the metal-support interaction and stability with respect to sintering has been investigated for Pt nanoparticles supported on well-ordered Co3O4(111)/Ir(100) films in UHV and under oxidizing conditions by means of synchrotron radiation photoelectron spectroscopy (SRPES) and near ambient pressure X-ray photoelectron spectroscopy (NAP XPS). The electronic metal-support interaction between Pt and Co3O4(111) associated with charge transfer results in partial reduction of Co3O4(111) yielding partially oxidized Ptδ+ species at the interface. The stability of the supported Pt particles is coupled with the oxidation state of Ptδ+ species, which can be reduced or oxidized depending on the Pt coverage and reactive environment. Annealing of Pt/Co3O4(111)/Ir(100) in UHV triggers the reduction of Ptδ+ species. At higher temperature, reverse spillover of oxygen to the Pt nanoparticles is accompanied by reduction of Co3O4(111). Under these conditions, the oxidation state of Ptδ+ species depends strongly on Pt coverage. Thus, at low Pt coverage (0.3 ML Pt), Ptδ+ is converted to Pt4+, at intermediate coverage (1.3 ML Pt), Ptδ+ remains stable, and at high Pt coverage (1.93 ML), Ptδ+ is reduced to Pt0. Sintering of Pt particles is associated with the reduction of the Ptδ+ species. This process is prevented under oxidizing conditions due to the formation of an interfacial oxide PtOx. The formation of an interfacial PtOx is observed at 300 K under exposure to 1 × 10−6 mbar O2 at Pt coverages below 1.3 ML. Using NAP XPS, we observe the formation of an interfacial PtOx at high Pt coverage (2.0 ML) in an oxygen atmosphere (1 mbar) at 300 K while the formation of surface PtOx is kinetically hindered and occurs above 550 K only.


 Please wait while we load your content...
Please wait while we load your content...