Anhydrous proton conduction in porous organic networks†
Abstract
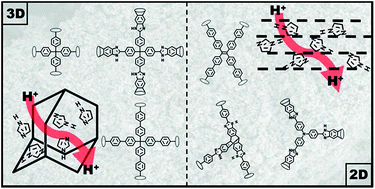
Solid electrolyte separators within fuel cells enable efficient charge transport and prevent a mass bypass between the two half cells. Hydrated systems, like Nafion, reach unprecedented proton conductivities at ambient temperatures, but the demanding humidity management prevents their use beyond 80 °C, hence limiting the efficiency of current polymer-based systems. As such, water free and chemically inert, solid materials with excellent conductivities between 100 °C and 200 °C, are of high interest. A promising approach is the incorporation of heavier amphoteric molecules into micro- and mesoporous frameworks. Stronger host–guest interactions allow for higher temperatures, while still maintaining sufficient mobility and efficient transport pathways. Here, we present a systematic study investigating the influence of porosity, framework topology and dimensionality as well as framework functionality and charge carrier uptake on the proton conductivity for six porous organic networks (PONs) loaded with imidazole via gas phase adsorption. The resulting materials were thoroughly characterized by multinuclear NMR and IR spectroscopy and physisorption as well as powder X-ray diffraction and DSC experiments, revealing homogeneous distribution of the amphoteric guests within the pore structure. Electrochemical impedance spectroscopy up to 130 °C revealed remarkable conductivities of up to 10−3 S cm−1 under anhydrous conditions. We found 3D networks to favour high imidazole loading leading to high proton conductivities based on the Grotthuss mechanism. In contrast, 2D networks showed a lower guest molecule uptake and thus lower proton conductivities, which were governed by vehicle transport. Additional acid/base functionalities within the frameworks seem to have a negative effect on the proton conduction.


 Please wait while we load your content...
Please wait while we load your content...