Robust FeCo nanoparticles embedded in a N-doped porous carbon framework for high oxygen conversion catalytic activity in alkaline and acidic media†
Abstract
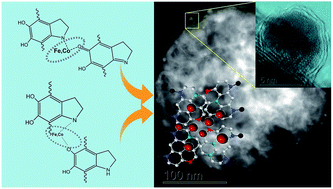
FeCo alloy nanoparticles were nucleated onto graphitic carbon layers through the pyrolysis of polydopamine (PDA) sub-micrometer spheres to form a highly active electrocatalytic system that exhibits excellent oxygen conversion catalytic activity in both alkaline and acidic media. Owing to the strong metal chelation capability during the chemical modification and high sp2-dominant carbon yield of PDA, an abundance of non-precious-metal ions were easily trapped and absorbed into the PDA segments at room temperature by catechol and amine functional groups, followed by the in situ nucleation of FeCo alloy nanoparticles on graphitic carbon layers during the pyrolysis. The contents of graphitic nitrogen and pyridinic nitrogen were significantly increased by the presence of the non-precious-metal ions during carbonization as well, which is a result of the chelation effect of non-precious-metal atoms. Meanwhile, the FeCo nanoparticles (diameter < 5 nm) were protected by the multi-layer-graphene-like carbon layer from the harsh acid and uniformly anchored on graphitic carbon sub-microspheres, which can greatly improve the catalytic durability, particularly in acidic media. From the perspective of the whole catalytic system being used as an air electrode in rechargeable Zn–air batteries, the porous nitrogen-doped graphitic carbon framework was functionalised as a continuous conductive framework due to its catalytic activity towards high oxygen conversion and good electrical conductivity.


 Please wait while we load your content...
Please wait while we load your content...