Aluminum enhances photochemical charge separation in strontium titanate nanocrystal photocatalysts for overall water splitting†
Abstract
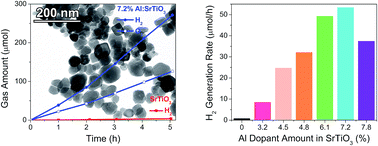
Strontium titanate (SrTiO3) is a well-known photocatalyst for overall water splitting (OWS) under ultra-violet irradiation. Recently, it was shown that Al-doped SrTiO3 microparticles prepared by flux-mediated solid-state reaction catalyze OWS with an apparent quantum efficiency of 30% at 360 nm, the highest value reported so far for strontium titanate. However, the roles of Al3+ and of the particle morphology in controlling the activity are not clear. Here we report a new synthetic route to well defined nanocrystals of Al:SrTiO3 with controllable Al-concentration (3–8 atom%) using TiO2, Sr(OH)2 and Al(NO3)3 as starting materials. X-Ray diffraction and transmission electron microscopy confirm the presence of perovskite-type cubic nanoparticles with size of 60 nm. After loading with a Rh2−yCryO3 cocatalyst, nano-Al:SrTiO3 catalyzes OWS under UV/vis illumination from a Xe lamp. The activity shows a volcano-like dependence on Al concentration, confirming that Al3+ content is a key factor for the activity. Nanocrystals doped with 7.2 atom% Al show the activity of 53 μmol H2 per hour under 240 mW cm−2 UV illumination and an AQE of 0.06% at 375 nm. Based on surface photovoltage spectroscopy (SPS), aluminum enhances photochemical charge separation in the Al:SrTiO3 nanocrystals and reduces electron and hole trapping. However, under UV/vis illumination, the Al:SrTiO3 nanocrystals are found to be 40 times less active than Al:SrTiO3 microparticles made by flux reaction in SrCl2. The lower activity of the nanoparticles is due to optical shielding from the Rh2−yCryO3 co-catalyst.


 Please wait while we load your content...
Please wait while we load your content...
