Nanostructured NiFe (oxy)hydroxide with easily oxidized Ni towards efficient oxygen evolution reactions†
Abstract
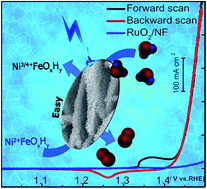
Electrochemical water splitting into hydrogen and oxygen is one of the most promising strategies for the utilization and storage of solar energy. However, the sluggish kinetics of the oxygen evolution reaction (OER) hinder its larger-scale application. NiFe-based catalysts are some of the most efficient oxygen evolution catalysts (OECs). High-valence Ni is considered as an active site or conductive framework in NiFe-based OECs. However, the oxidation of Ni is usually hindered in the presence of Fe. This paper describes an effective strategy to produce NiFe (oxy)hydroxide with easily oxidized Ni and a hierarchical nanosheet structure as the most efficient OEC. High valence Ni may provide frameworks with good conductivity and facilitate OERs in Fe active sites. In addition, the hierarchical structure can increase the electrochemical surface area, resulting in more active sites for the OER. The as-prepared Fe–O–Ni(OH)2 supported on nickel foam needs overpotentials of 185, 220 and 261 mV to drive current densities of 10, 100 and 500 mA cm−2, respectively. The catalyst also shows excellent stability at the current densities of 100 and 500 mA cm−2 for 50 hours.


 Please wait while we load your content...
Please wait while we load your content...