Core–shell CuPd@Pd tetrahedra with concave structures and Pd-enriched surface boost formic acid oxidation†
Abstract
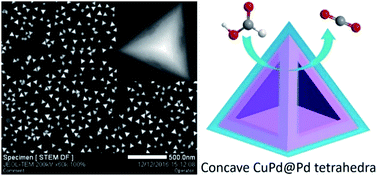
The shape control of multi-metal nanocrystals with concave structures is significant for constructing high-efficiency electrocatalysts. Herein, we report an effective one-pot hydrothermal synthetic strategy that allows high-yield production of CuPd@Pd core–shell tetrahedra with concave and Pd-enriched surfaces. It is demonstrated that L-proline plays a vital role as a structure-directing agent in tuning the nucleation and growth of concave CuPd@Pd tetrahedra because of the strong coordination abilities of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, O–H and N–H groups with metal ions. This bimetallic concave structure can endow the nanocrystals with abundant catalytically active sites and fascinating electronic effects, which are suggested to be favorable for electrocatalysis. When used as an electrocatalyst, the newly developed catalyst presented 4.2 and 2.4 times enhanced mass activity (501.8 A gPd−1) and specific activity (49.3 A m−2) for the formic acid oxidation when compared with those of the commercial Pd black catalyst (120.6 A gPd−1; 20.8 A m−2), respectively. Moreover, the concave CuPd@Pd catalyst exhibits substantially enhanced electrocatalytic stability under harsh electrochemical conditions and superior resistance to COads poisoning; this demonstrates that it is a promising candidate as an anode catalyst in direct formic acid fuel cells.
O, O–H and N–H groups with metal ions. This bimetallic concave structure can endow the nanocrystals with abundant catalytically active sites and fascinating electronic effects, which are suggested to be favorable for electrocatalysis. When used as an electrocatalyst, the newly developed catalyst presented 4.2 and 2.4 times enhanced mass activity (501.8 A gPd−1) and specific activity (49.3 A m−2) for the formic acid oxidation when compared with those of the commercial Pd black catalyst (120.6 A gPd−1; 20.8 A m−2), respectively. Moreover, the concave CuPd@Pd catalyst exhibits substantially enhanced electrocatalytic stability under harsh electrochemical conditions and superior resistance to COads poisoning; this demonstrates that it is a promising candidate as an anode catalyst in direct formic acid fuel cells.


 Please wait while we load your content...
Please wait while we load your content...