Effect of glycine functionalization of 2D titanium carbide (MXene) on charge storage†
Abstract
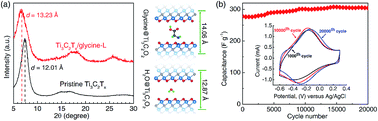
Restacking of two-dimensional (2D) flakes reduces the accessibility of electrolyte ions and is a problem in energy storage and other applications. Organic molecules can be used to prevent restacking and keep the interlayer space open. Here, we report on a combined theoretical and experimental investigation of the interaction between 2D titanium carbide (MXene), Ti3C2Tx, and glycine. From first principle calculations, we presented the functionalization of glycine on the Ti3C2O2 surface, evidenced by the shared electrons between Ti and N atoms. To experimentally validate our predictions, we synthesized flexible freestanding films of Ti3C2Tx/glycine hybrids. X-ray diffraction and X-ray photoelectron spectroscopy confirmed the increased interlayer spacing and possible Ti–N bonding, respectively, which agree with our theoretical predictions. The Ti3C2Tx/glycine hybrid films exhibited an improved rate and cycling performances compared to pristine Ti3C2Tx, possibly due to better charge percolation within expanded Ti3C2Tx.


 Please wait while we load your content...
Please wait while we load your content...