A microporous metal–organic framework with commensurate adsorption and highly selective separation of xenon†
Abstract
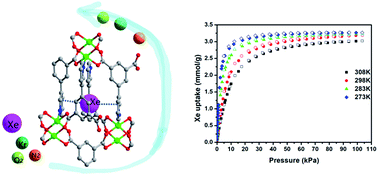
The separation of xenon (Xe) and krypton (Kr) becomes increasingly important due to the industrial significance of high-purity Xe gas and the concern with reprocessing radioactive isotopes of Xe and Kr at parts per million concentrations from the off-gas of used nuclear fuel. Current separation processes mainly rely on energy and capital intensive cryogenic distillation. Thus, more economical and energy-efficient alternatives, such as physisorptive separation, using porous materials are needed to be developed. Herein, we present a microporous metal–organic framework (MOF-Cu-H) in which the suitable pore/cage-like structure with a precise size matching with the xenon atom leads to its commensurate adsorption phenomenon of Xe under ambient conditions and superior performance for Xe capture and separation. MOF-Cu-H exhibits by far the highest Xe Henry coefficient, remarkable Xe/Kr selectivity and significantly high Xe adsorption capacity at very low partial pressures relevant to nuclear fuel reprocessing. Temperature dependent isotherms, adsorption kinetics experiments, single column breakthrough curves and molecular simulation studies collaboratively support the claim, underlining the potential of this material for energy and cost-effective removal of xenon from nuclear fuel reprocessing plants compared with cryogenic distillation.


 Please wait while we load your content...
Please wait while we load your content...