Oxygen redox in hexagonal layered NaxTMO3 (TM = 4d elements) for high capacity Na ion batteries†
Abstract
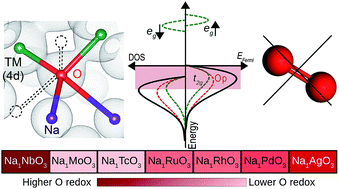
Through comprehensive density functional calculations, we demonstrate oxygen's significant participation in the redox reaction in a Na excess NaxRuO3 cathode material. The availability of O electrons for the redox reaction originates from the local coordination environment. For high sodium content (x ≈ 2), O ions in the layered hexagonal Na2RuO3 compound are coordinated by four Na ions and consequently have their 2p electrons lifted closer to the Fermi level. For lower Na content (x ≈ 1), Na1RuO3 adopts an ilmenite type R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) structure in which O ions are coordinated by two Ru and two Na ions. In this case, O under-coordination further elevates O 2p states closer to the Fermi level. In both cases, high O electronic population near the Fermi level facilitates continuous participation of O in the redox reaction over a wide range of Na concentrations. Based on this concept, we also predict that Na1NbO3 with an ilmenite framework is a suitable and economical candidate for high voltage and high capacity cathodes for Na ion batteries.
structure in which O ions are coordinated by two Ru and two Na ions. In this case, O under-coordination further elevates O 2p states closer to the Fermi level. In both cases, high O electronic population near the Fermi level facilitates continuous participation of O in the redox reaction over a wide range of Na concentrations. Based on this concept, we also predict that Na1NbO3 with an ilmenite framework is a suitable and economical candidate for high voltage and high capacity cathodes for Na ion batteries.


 Please wait while we load your content...
Please wait while we load your content...