Monolayer InP3 as a reversible anode material for ultrafast charging lithium- and sodium-ion batteries: a theoretical study
Abstract
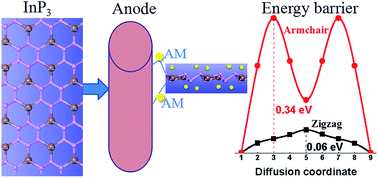
Improving the battery capacity usually induces a large volume expansion of the electrode, leading to poor reversibility and safety of the battery. Therefore, high specific capacity, fast charging rate, and good reversibility should be balanced in electrode materials. Using first-principles calculations, we systematically explore the InP3 monolayer as an anode material to hold Li (Na) atoms. The adsorption of Li (Na) induces the transformation of the sp2 hybridization of In to the sp3 hybridization, implying a strong binding and fast loading of Li (Na). Furthermore, the alkali-metal–InP3 system shows metallic characteristics which give rise to good electrical conductivity. In addition, Li (Na) diffusion in the zigzag direction is faster than that along the armchair direction. Especially, the Na diffusion barrier along the zigzag direction is as low as 0.06 eV, corresponding to ultrafast charge/discharge capability. Balancing the stability and storage capacity, the specific capacity and average electrode potential of Li (Na) are 258.1 (258.1) mA h g−1 and 0.53 (1.49) V, respectively. Finally, the structure of InP3 is well maintained upon the adsorption of Li (Na) even at the maximum concentration, suggesting its good stability and reversibility. The above-mentioned excellent properties suggest that the InP3 monolayer is a promising anode material for alkali-metal batteries.


 Please wait while we load your content...
Please wait while we load your content...