Local structure and conductivity behaviour in Bi7WO13.5†
Abstract
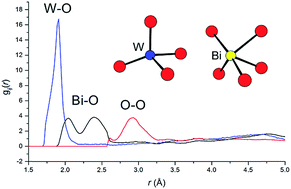
The structure and electrical properties of the oxide ion conductor Bi7WO13.5 have been examined as a function of temperature using neutron diffraction, differential thermal analysis and a.c. impedance spectroscopy. At room temperature, Bi7WO13.5 exhibits the type Ib tetragonally ordered fluorite structure, with tungsten distributed over two of the six cation sites. Reverse Monte Carlo (RMC) analysis of total neutron scattering data reveals a more realistic picture of the tungsten coordination environment than obtained by the average crystallographic analysis, with tungsten in predominantly tetrahedral coordination and an average bismuth coordination number of around 5. At 893 °C on heating Bi7WO13.5 undergoes a phase transition to a cubic δ-Bi2O3 type phase. The total scattering analysis of this phase reveals significant detail of the local environments of the cations. In particular, a bimodal distribution is found for Bi–O correlations, which has been used to guide the crystallographic analysis. Even in the type Ib phase Bi7WO13.5 shows relatively high conductivity in the intermediate temperature range and measurements of the oxide ion transference number confirm this to be virtually pure ionic in nature. Analysis of the oxide ion vacancy distributions shows a preference for 〈100〉 vacancy pair alignment in both phases. Evidence for a second order phase transition at around 400 °C is discussed.
- This article is part of the themed collection: Advances in Solid State Chemistry and its Applications


 Please wait while we load your content...
Please wait while we load your content...