Carbon–TiO2 composites as high-performance supercapacitor electrodes: synergistic effect between carbon and metal oxide phases†
Abstract
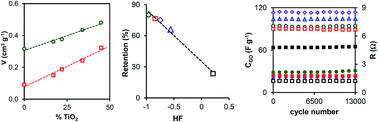
A series of carbon xerogels doped with different percentages of TiO2 has been studied as a tentative means of preparing electrodes for supercapacitors. Carbon composites were obtained by an inverse emulsion method in n-heptane, and after carbonization at 900 °C the metal oxide phase was well dispersed in the carbon phase with a crystal size of less than 4 nm, and only an anatase phase was detected. An increase in the percentage of TiO2 produced a decrease in the hydrophobicity of the composite, which improved the wettability of the electrodes. XPS results showed that Ti3+ and Ti4+ were present on the surface of samples, and the presence of both oxidation states can improve the electron mobility in the inorganic phase. The obtained composite materials possessed specific surface areas that ranged from 423 to 539 m2 g−1 and very well developed micro- and mesoporosity with a total pore volume that ranged from 0.361 to 0.480 cm3 g−1. The mean size of supermicropores increased as the percentage of TiO2 increased, whereas practically no variation was found in the size of ultramicropores. Two-electrode symmetric supercapacitors based on the carbon xerogel–TiO2 composites exhibited high electrochemical performance, which was better than that of other similar materials in the literature, and displayed high capacitance (up to 137 F g−1 at 0.250 A g−1 for the composite containing 20% TiO2), high capacitance retention (66–80%) at 20 A g−1 and a high energy density of 20.15 W h kg−1 at a power density of 138.11 W kg−1 in the voltage range of 0 V to 1.1 V. A sample with a combination of low hydrophobicity and an adequate micro/mesopore network with an intermediate content of TiO2 exhibited the best performance for energy storage. A floating test showed the very good cyclability of the synthesized materials.


 Please wait while we load your content...
Please wait while we load your content...