Achieving convenient CO2 electroreduction and photovoltage in tandem using potential-insensitive disordered Ag nanoparticles†
Abstract
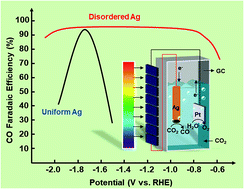
Photovoltaic-electrochemical (PV-EC) systems can not only make full use of solar energy, but also transform CO2 into organic molecules. However, it is difficult to achieve PV-EC systems since most CO2 reduction catalysts are potential-dependent. This paper describes the rational design of potential-insensitive disordered Ag, which can achieve more than 90% faradaic efficiency (FE) for CO within a wide voltage range of 1.1 V in an electroreduction CO2 system. The system shows attractive activity under different photovoltage conditions in a PV-EC system. By employing in situ attenuated total reflection surface-enhanced infrared absorption spectroscopy (ATR-SEIRAS), we address the origin of the volcano peak of FE on Ag nanoclusters to understand the mechanism of the carbon dioxide reduction reaction (CO2RR). In addition, we find that the CO2RR on disordered Ag nanoparticles is a proton–electron coupling transfer (PECT) reaction mechanism, which may result in high activity in a wide potential range.
- This article is part of the themed collection: Editor’s Choice – Jihong Yu


 Please wait while we load your content...
Please wait while we load your content...