Rechargeable aluminum–selenium batteries with high capacity†
Abstract
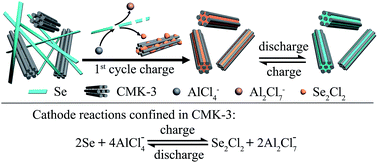
Rechargeable aluminum (Al) batteries are emerging as a promising post lithium-ion battery technology. Herein, we demonstrate a conceptually new design of rechargeable aluminum–selenium (Al–Se) batteries by understanding the selenium chemistry and controlling the electrode reaction. The Al–Se battery consists of a composite cathode including selenium nanowires and mesoporous carbon (CMK-3) nanorods, an Al metal anode and chloroaluminate ionic liquid electrolyte. The working mechanism of the Al–Se battery is the reversible redox reaction of the Se2Cl2/Se pair confined in the mesopores of CMK-3 nanorods. Al–Se batteries deliver a high reversible capacity of 178 mA h g−1 (by Se mass), high discharge voltages (mainly above 1.5 V), and good cycling/rate performances.
- This article is part of the themed collection: 2018 Chemical Science HOT Article Collection


 Please wait while we load your content...
Please wait while we load your content...