A novel resource utilization method using wet magnesia flue gas desulfurization residue for simultaneous removal of ammonium nitrogen and heavy metal pollutants from vanadium containing industrial wastewater
Abstract
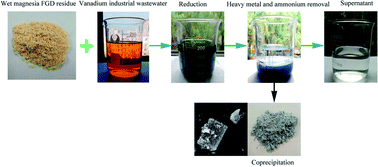
In the present study, a novel resource utilization method using wet magnesia flue gas desulfurization (FGD) residue for the simultaneous removal of ammonium nitrogen (NH4–N) and heavy metal pollutants from vanadium (V) industrial wastewater was proven to be viable and effective. In this process, the wet magnesia FGD residue could not only act as a reductant of hexavalent chromium [Cr(VI)] and pentavalent vanadium [V(V)], but also offered plenty of low cost magnesium ions to remove NH4–N using struvite crystallization. The optimum experimental conditions for Cr(VI) and V(V) reduction are as follows: the reduction pH = 2.5, the wet magnesia FGD residue dose is 42.5 g L−1, t = 15.0 min. The optimum experimental conditions for NH4–N and heavy metal pollutants removal are as follows: the precipitate pH = 9.5, the n(Mg2+) : n(NH4+) : n(PO43−) = 0.3 : 1.0 : 1.0, t = 20.0 min. Finally the NH4–N, V and Cr were separated from the vanadium containing industrial wastewater by forming the difficult to obtain, soluble coprecipitate containing struvite and heavy metal hydroxides. The residual pollutant concentrations in the wastewater were as follows: Cr(VI) was 0.047 mg L−1, total Cr was 0.1 mg L−1, V was 0.14 mg L−1, NH4–N was 176.2 mg L−1 (removal efficiency was about 94.5%) and phosphorus was 14.7 mg L−1.


 Please wait while we load your content...
Please wait while we load your content...