Sulfur compounds reactivity in the ODS of model and real feeds on W–SBA based catalysts†
Abstract
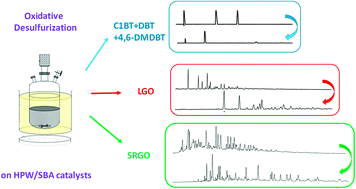
W based catalysts were synthesized by dry impregnation of SBA-15 mesoporous silica with phosphotungstic acid (HPW) solution with W contents between 5 and 20%, the HPW compound being preserved after calcination. The catalysts performance and the reactivity of various sulfide compounds were evaluated in the oxidative desulfurization (ODS) of model solutions and of real diesels, with sulfur contents ranging from 50 to 2000 ppm. The reactivity of benzothiophene and dibenzothiophene compounds was different in the ODS of model solutions but globally identical in the ODS of SRGO. The monitoring of the concentration of a range of alkyl DBT compounds (with alkyl groups from C2 to C5) in LGO confirmed the importance of the steric hindrance of alkyl substituents in the 4,6 position near the S atom, as well as of the size of the alkyl groups. Among the xW/SBA series, the catalyst with the highest loading showed the best performance in the ODS of LGO and SRGO while the catalysts efficiency could not be discriminated in the ODS of model solutions. In the ODS of both model solutions and real feeds, the W/SBA catalyst was found to be much more efficient than a catalyst obtained by impregnation of a commercial silica with similar loading, highlighting the beneficial use of a mesoporous support with high surface area and pore volume that allowed well-dispersed tungsten species to be obtained. The quantity of sulfones precipitated and/or retained on the catalyst depended on the feed and was found to be higher in the ODS of model solution than in the ODS of real feeds. The precipitated/retained sulfones on the support may induce catalyst deactivation, which highlights the importance of the textural properties of the support. This detailed study points out the difficulty of extrapolating results obtained in the ODS of model solution to the ODS of real feeds.


 Please wait while we load your content...
Please wait while we load your content...