17O solid-state NMR spectroscopy of A2B2O7 oxides: quantitative isotopic enrichment and spectral acquisition?†
Abstract
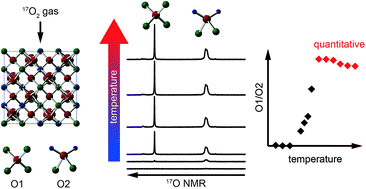
The potential of 17O NMR spectroscopy for the investigation of A2B2O7 ceramic oxides important in the encapsulation of radioactive waste is demonstrated, with post-synthetic enrichment by exchange with 17O2 gas. For Y2Sn2O7, Y2Ti2O7 and La2Sn2O7 pyrochlores, enrichment of the two distinct O species is clearly non quantitative at lower temperatures (∼700 °C and below) and at shorter times, despite these being used in prior work, with preferential enrichment of OA2B2 favoured over that of OA4. At higher temperatures, the 17O NMR spectra suggest that quantitative enrichment has been achieved, but the integrated signal intensities do not reflect the crystallographic 1 : 6 (O1 : O2) ratio until corrected for differences in T1 relaxation rates and, more importantly, the contribution of the satellite transitions. 17O NMR spectra of Y2Zr2O7 and Y2Hf2O7 defect fluorites showed little difference with any variation in enrichment temperature or time, although an increase in the absolute level of enrichment (up to ∼7.5%) was observed at higher temperature. DFT calculations show that the six distinct resonances observed cannot be assigned unambiguously, as each has contributions from more than one of the five possible next nearest neighbour environments. For La2Ti2O7, which adopts a layered perovskite-like structure, little difference in the spectral intensities is observed with enrichment time or temperature, although the highest absolute levels of enrichment (∼13%) were obtained at higher temperature. This work demonstrates that 17O NMR has the potential to be a powerful probe of local structure and disorder in oxides, but that considerable care must be taken both in choosing the conditions for 17O enrichment and the experimental acquisition parameters if the necessary quantitative measurements are to be obtained for more complex systems.


 Please wait while we load your content...
Please wait while we load your content...