A correlation study of biological activity and molecular docking of Asp and Glu linked bis-hydrazones of quinazolinones†
Abstract
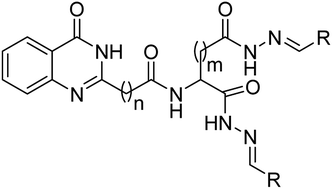
The present investigation involves the synthesis and spectroscopic and biological activity studies of the bis-hydrazones of quinazolinones derived from aspartic acid and glutamic acid. The antioxidant activities of the compounds were evaluated using DPPH, DMPD and ABTS radical scavenging assays whose results revealed that the IC50 of compounds 6, 7, 11, 12, 20, 21, 25 and 26 was lower than those of the standard references. The anti-inflammatory activity was evaluated with a haemolysis assay using a human blood erythrocytes suspension and the results demonstrated that compounds 8, 9, 13, 14, 22, 23, 27 and 28 were excellent anti-inflammatory agents. In addition, the antibacterial and antifungal activities against various clinical pathogens of human origin revealed that compounds 7, 9, 12, 14, 21, 23, 26 and 28 possessed potent antimicrobial properties. Furthermore, to understand the correlation between biological activity and drug–receptor interaction, molecular docking was performed on the active sites of tyrosine kinase (PDB ID: 2HCK), cyclooxygenase-2 (PDB ID: 1CX2) and glucosamine-6-phosphate (GlcN-6-P) synthase (PDB ID: 2VF5) which showed good binding profiles with the targets that can potentially hold the title compounds. The correlation study revealed that compounds containing EDGs (–OH, –OCH3) were excellent antioxidants, compounds with EWGs (–Cl, –NO2) exhibited good anti-inflammatory activity and compounds bearing –OH and –NO2 groups were very good antimicrobials.


 Please wait while we load your content...
Please wait while we load your content...