C2H5OH and NO2 sensing properties of ZnO nanostructures: correlation between crystal size, defect level and sensing performance
Abstract
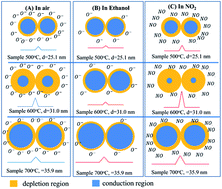
ZnO nanostructures can be synthesized using different techniques for gas sensor applications, but different synthesis methods produce different morphologies, specific surface areas, crystal sizes, and physical properties, which consequently influence the gas-sensing properties of materials. Many parameters such as morphology, specific surface areas, crystal sizes, and defect level can influence the gas-sensing properties of ZnO nanostructures. However, it is not clear which parameter dominates the gas-sensing performance. This study clarified the correlation between crystal size, defect level, and gas-sensing properties of ZnO nanostructures prepared from hydrozincite counterparts by means of field emission scanning electron microscopy, high resolution transmission electron microscopy, X-ray diffraction and photoluminescence spectra. Results showed that the average crystal size of the ZnO nanoparticles increased with thermal decomposition temperatures from 500 °C to 700 °C. However, the sample treated at 600 °C, which has the lowest visible-to-ultraviolet band intensity ratio showed the highest response to ethanol and NO2. These results suggested that defect level but not size is the main parameter dominating the sensor performance. The gas sensing mechanism was also elucidated on the basis of the correlation among decomposition temperatures, crystal size, defect level, and gas sensitivity.


 Please wait while we load your content...
Please wait while we load your content...