Photocatalytic activity of SnO2–α-Fe2O3 composite mixtures: exploration of number of active sites, turnover number and turnover frequency†
Abstract
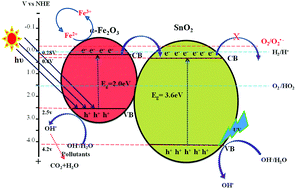
SnO2 and α-Fe2O3 were prepared by a sol–gel method and their composite (SnO2–α-Fe2O3) systems were synthesized by combining SnO2 with α-Fe2O3 in various weight percent ratios. Sn : Fe (0.25 : 0.75), Sn : Fe (0.5 : 0.5) and Sn : Fe (0.75 : 0.25) samples were prepared using a simple grinding process and they were characterized by various analytical techniques. The photocatalytic activity was studied under UV/solar light using phenol as a model pollutant. The better performance of the Sn : Fe (0.75 : 0.25) composite under UV/solar light irradiation was due to the efficient charge carrier separation and also due to the synergistic effects between photocatalysis and photo-Fenton reactions. The Sn : Fe (0.25 : 075) composite shows better photocatalytic activity under solar light illumination. The photocatalytic activity of the composite systems further increases in the presence of H2O2. An extremely small amount of the catalyst (10 mg/250 mL) was capable of causing a considerable increase in the rate of the degradation reaction. The effectiveness of the catalyst at very low concentrations is suggested by the calculation of number of active sites, turnover number (TON) and turnover frequency (TOF) for all the systems under both UV/solar light illumination. The photocatalytic oxidation process dominates the degradation reaction rather than the photocatalytic reduction. The staggered positions of the conduction band edges of SnO2 and α-Fe2O3 with respect to one another facilitate a smooth charge transfer process.


 Please wait while we load your content...
Please wait while we load your content...