From mono- to tetraacylgermanes: extending the scope of visible light photoinitiators†
Abstract
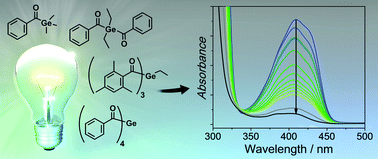
We have investigated the inititiaton efficiency of carefully selected germanium-based photointiators for radical polymerization. To establish a systematic relationship between structure and reactivity, we have developed a convenient synthetic protocol for the preparation of a trisacylgermane, closing the gap from mono- to tetraacylgermane photoinitiators. The studied acylgermanes display distinct, wavelength-dependent photobleaching upon irradiation up to 470 nm. In particular, tetraacylgermanes featuring ortho-alkyl substituents reveal red-shifted n–π* bands, in line with excellent photobleaching upon visible light irradiation. Quantum yields of decomposition (determined at 385 nm) have been found to be highest for bisacylgermanes. Germyl radicals produced upon triplet-state α-cleavage of the acylgermanes react remarkably fast with monomers. Addition rate constants to (meth)acrylates range from 0.4–4.5 × 108 M−1 s−1, depending on the substitution pattern. These values are clearly higher than those reported for related phosphorus-centered radicals derived from acylphosphane oxides. We have further established the nature of the products and side-products formed at initial stages of the polymerizations using chemically induced dynamic nuclear polarization (CIDNP) experiments.


 Please wait while we load your content...
Please wait while we load your content...