β-Diketone derivatives: influence of the chelating group on the photophysical and mechanofluorochromic properties†
Abstract
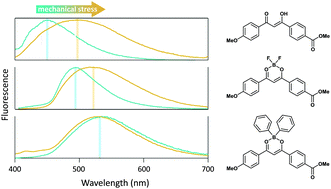
A diphenyl-boron β-diketonate complex was synthesized. Its photophysical properties were studied in solution and in the solid-state, and compared to those of its parent diketone and the corresponding difluoro-boron complex. TD-DFT calculations show that the molecular orbitals involved in the first Franck–Condon transition are very different for the three compounds studied. The difluoro-boron complex is strongly fluorescent in solution, and remains fluorescent in the solid-state. The free diketone turns to be very weakly fluorescent in solution and displays significant Aggregation Induced Enhanced Emission (AIEE) in the crystalline state, which can be explained by a rigidification of the molecule, while the diphenyl-boron complex is weakly fluorescent in solution as well as in the solid-state. For the free diketone and the difluoro-boron complex a mechanofluorochromic response is observed upon grinding the crystalline powder in a mortar, while for the diphenyl-boron complex no fluorescence emission change is detected under these conditions. Overall, this study shows that the nature of the chelating group has a crucial influence on the photophysical and mechanofluorochromic properties of β-diketonate complexes, leading to a wide variety of behaviors within the closely related structures of such derivatives.
- This article is part of the themed collection: 28th International Conference on Photochemistry (ICP 2017)


 Please wait while we load your content...
Please wait while we load your content...