Chemoenzymatic macrocycle synthesis using resorcylic acid lactone thioesterase domains†
Abstract
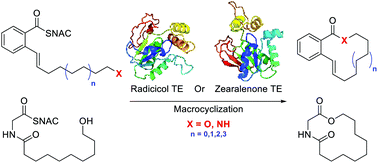
A key missing tool in the chemist's toolbox is an effective biocatalyst for macrocyclization. Macrocycles limit the conformational flexibility of small molecules, often improving their ability to bind selectively and with high affinity to a target, making them a privileged structure in drug discovery. Macrocyclic natural product biosynthesis offers an obvious starting point for biocatalyst discovery via the native macrocycle forming biosynthetic mechanism. Herein we demonstrate that the thioesterase domains (TEs) responsible for macrocyclization of resorcylic acid lactones are promising catalysts for the chemoenzymatic synthesis of 12- to 18-member ring macrolactones and macrolactams. The TE domains responsible for zearalenone and radicicol biosynthesis successfully generate resorcylate-like 12- to 18-member macrolactones and a 14-member macrolactam. In addition these enzymes can also macrolactonize a non-resorcylate containing depsipeptide, suggesting they are versatile biocatalysts. Simple saturated omega-hydroxy acyl chains are not macrocyclized, nor are the alpha-beta unsaturated derivatives, clearly outlining the scope of the substrate tolerance. These data dramatically expand our understanding of substrate tolerance of these enzymes and are consistent with our understanding of the role of TEs in iterative polyketide biosynthesis. In addition this work shows these TEs to be the most substrate tolerant polyketide macrocyclizing enzymes known, accessing resorcylate lactone and lactams as well as cyclicdepsipeptides, which are highly biologically relevant frameworks.
- This article is part of the themed collections: Synthetic methodology in OBC, Catalysis & biocatalysis in OBC and Supramolecular chemistry in OBC


 Please wait while we load your content...
Please wait while we load your content...