Copper-catalyzed generation of flavone selenide and thioether derivatives using KSeCN and KSCN via C–H functionalization†
Abstract
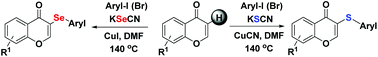
Flavone selenide or sulfur-containing derivatives are pretty valuable in drug discovery due to their diversity of important bioactivities. Here, two simple Cu-catalyzed methods of constructing C–Se and C–S bonds on flavone skeletal structures via C–H functionalization are reported, which regioselectively afford flavone selenide and sulfide derivatives in good yields using cheap KSeCN or KSCN salts as selenium and sulfur agents. These methods further enrich the current C–Se and C–S bond construction methods.
- This article is part of the themed collection: Synthetic methodology in OBC


 Please wait while we load your content...
Please wait while we load your content...